Article Text
Abstract
Background Pain management after total knee arthroplasty is essential to improve early mobilization, rehabilitation, and recovery. Continuous adductor canal (AC) block provides postoperative analgesia while preserving quadriceps strength. However, there have been inconsistencies regarding the optimal location for continuous catheter block. We compared continuous femoral triangle, proximal AC, and distal AC blocks for postoperative analgesia after total knee arthroplasty.
Methods Patients undergoing unilateral total knee arthroplasty were randomly assigned to three groups: femoral triangle, proximal AC, or distal AC. The surgeon performed periarticular local anesthetic infiltration. After surgery, an ultrasound-guided perineural catheter insertion procedure was performed. The primary endpoint was pain scores at rest in the morning on the first postoperative day. Secondary endpoints included pain scores at rest and during activity at other time points, quadriceps strength, and opioid consumption.
Results Ninety-five patients, 32 in the femoral triangle group, 31 in the proximal AC group, and 32 in the distal AC group, completed the study. Analysis of the primary outcome showed no significant difference in pain scores among groups. Secondary outcomes showed significantly lower pain scores at rest and during activity in the distal AC group than in the femoral triangle and proximal AC groups in the morning of the second postoperative day. Quadriceps strength and opioid consumption did not differ among groups.
Conclusions Continuous femoral triangle, proximal AC, and distal AC blocks in the setting of periarticular local anesthetic infiltration provide comparable postoperative analgesia after total knee arthroplasty.
Trial registration number NCT04206150.
- analgesia
- nerve block
- pain, postoperative
- lower extremity
Data availability statement
Data are available upon reasonable request.
Statistics from Altmetric.com
Introduction
Total knee arthroplasty (TKA) is associated with moderate to severe pain in 50% of patients in the first three postoperative days.1 Optimal pain management after TKA is therefore essential to improve early mobilization, rehabilitation, and recovery.2 As a key component of effective multimodal analgesia, continuous adductor canal block (ACB) provides postoperative analgesia while preserving quadriceps strength.3
The adductor canal (AC) is a triangular musculoaponeurotic conduit between the apex of the femoral triangle and adductor hiatus. Previous clinical studies on continuous ACBs used surface anatomical landmarks to identify the proximal end of the AC as the midpoint between the anterior superior iliac spine (ASIS) and the superior border of the patella.4 5 Recent cadaveric studies have revealed that the proximal end of the AC is located distal from the midpoint of the thigh.6–8 In recent clinical studies regarding ACB catheter placement, the proximal end of the AC was precisely identified using ultrasound landmarks as the intersection of the medial borders of the sartorius muscle and the adductor longus muscle.9–11 However, despite previous studies, the ideal location of continuous ACB catheter analgesia after TKA has been difficult to determine due to inconsistent AC identification and various catheter insertion positions.4 5 9–11 In addition, most studies have compared femoral triangle block with ACB for continuous catheter analgesia, showing inconsistent results.5 9 10 Therefore, a comparison of analgesic efficacy provided by catheters inserted into the femoral triangle, proximal AC, and distal AC may provide an objective basis for a clear recommendation on this approach.
We designed this randomized, controlled, double-blind clinical trial to compare the effects of continuous femoral triangle, proximal AC, and distal AC blocks on postoperative analgesia after TKA. Our primary endpoint was to compare pain scores in the morning on postoperative day 1 (POD1). The secondary endpoints included pain scores at other time points, quadriceps muscle strength, and opioid consumption.
Methods
This randomized, controlled, double-blind study was registered at ClinicalTrials.gov on December 20, 2019. A total of 96 adult patients with an American Society of Anesthesiologists physical status of class I–III scheduled for elective, unilateral, primary TKA were enrolled between April 2020 and July 2021. Exclusion criteria were chronic opioid use (daily use for >1 month of 20 mg oral morphine equivalents), revision surgery, contraindications to spinal anesthesia or peripheral nerve blocks, and allergy to lidocaine or ropivacaine. Written informed consent was obtained from all patients.
Patients were randomly assigned to either the femoral triangle, proximal AC, or distal AC groups according to a computer-generated randomization sequence on the day of surgery. Patient allocation was performed by an investigator who was not involved in postoperative assessments. Randomization assignments were concealed until catheter insertion. Aside from the investigator performing the catheter insertion procedure, other investigators, surgeons, patients, attending anesthesiologists, and nurses were blinded to group assignments during the study period.
Anesthesia and surgery protocol
On the patients’ arrival at the operating room, routine monitoring was performed. The patients received spinal anesthesia with 10–12 mg of 0.5% hyperbaric bupivacaine. All TKAs were performed via the medial parapatellar approach by a single surgical team. The surgeon performed periarticular local anesthetic infiltration with 150 mg of ropivacaine and 30 mg of ketorolac diluted with normal saline to a total volume of 50 mL. The first 20 mL of the mixture were injected into the posterior capsule, the medial and lateral collateral ligaments, and medial and lateral meniscus remnants, prior to implant placement. The remaining 30 mL were used to infiltrate the soft tissue around the knee, including the surrounding muscles, synovium, fat pad, and subcutaneous tissue, before wound closure. All patients received preoperative celecoxib (200 mg) orally and intraoperative doses of acetaminophen (1 g), tranexamic acid (1 g), and dexamethasone (5 mg) intravenously. In the ward, all patients received celecoxib 200 mg orally and acetaminophen 1 g intravenously every 12 hours thereafter. Intravenous tramadol (50 mg) was administered as rescue analgesia. Patients received standard of care physical therapy sessions (two times on POD1 and POD2) and were mobilized once on the day of surgery with caregiver assistance and ambulated as soon as possible on POD1.
Catheter insertion procedure
After surgery, the patient was transferred to a block room and routine monitoring was performed. All catheters were inserted under a full aseptic technique using a real-time ultrasound-guided in-plane approach by an experienced anesthesiologist. A multi-hole perineural catheter through a catheter-over-needle system (E-cath PLUS, PAJUNK GmbH, Geisingen, Germany) and a linear 6–13-MHz ultrasound probe (HFL38xp, SonoSite Inc., Bothell, Washington, USA) were used for all patients.
On ultrasound, the proximal end of the AC was defined as the intersection of the medial borders of the sartorius muscle and the adductor longus muscle (online supplemental figure 1). In the femoral triangle group, the insertion site was determined at 2/15th the femur length above the proximal end of the AC along the long axis of the femur (figure 1A). In the proximal AC group, the insertion site was the proximal end of the AC (figure 1B). In the distal AC group, the insertion site was determined at 1/15th the femur length below the proximal end of the AC along the long axis of the femur (figure 1C). In our hospital’s TKA protocol, a full-length standing anteroposterior radiograph is obtained before admission. Based on this, femur length was measured from the top of the femoral head to the most distal end of the medial femoral condyle (online supplemental figure 2).12
Supplemental material
Supplemental material
Location of catheter insertion and corresponding ultrasound images. (A) The probe moves cranially along the long axis of the thigh as 2/15th of the femur length from the proximal end of the AC. (B) The probe is positioned to identify the proximal end of the AC, where the medial borders of ALM and the sartorius muscle intersect. (C) The probe moves caudally along the long axis of the thigh as 1/15th of the femur length from the proximal end of the AC. AC, adductor canal; ALM, adductor longus muscle; AMM, adductor magnus muscle; FA, femoral artery; FV, femoral vein; SN, saphenous nerve; VMM, vastus medialis muscle.
In preliminary data from 10 patients, the mid-thigh was approximated at 2/15th of the femur length above the proximal end of the AC (online supplemental table 1). In addition, an area 2 cm above the adductor hiatus was approximated at 1/15th of the femur length below the proximal end of the AC. The proximal end of the AC was identified by ultrasound and the length from the ASIS to the superior border of the patella was measured using a tape measure.
Supplemental material
Following identification of the designated site, an 18-gage cannula with an indwelling 21-gage needle was advanced through the sartorius muscle with the needle tip positioned lateral to the saphenous nerve. To open a space for catheter insertion, 10 mL of 0.2% ropivacaine was injected via the needle for hydrodissection. A 21-gage multi-hole E-catheter was inserted through an indwelling 18-gage cannula. An additional 5 mL of 0.2% ropivacaine was injected during ultrasound imaging to ensure correct placement of the catheter tip between the artery and the deep fascia of the sartorius in the femoral triangle group or between the artery and the vastoadductor membrane (VAM) in the AC groups (online supplemental figure 3). The catheter was secured with a sterile occlusive dressing and an anchoring device. During the 48-hour postoperative period, 0.2% ropivacaine was infused via the perineural catheter at a basal rate of 6 mL/hour, a 4 mL bolus, and a lockout time of 30 min using a portable, patient-controlled infusion pump. After the procedure, all patients were dressed with hospital uniform tops and bottoms to blind the group assignment.
Supplemental material
Outcome measurement
The primary endpoint was pain scores at rest in the morning on POD1. The pain intensity at rest and during activity was evaluated using an 11-point numeric rating scale (NRS: 0=no pain, 10=worst imaginable pain). The secondary endpoints included pain scores at other time points, quadriceps muscle strength, opioid consumption, and local anesthetic consumption. Pain scores and quadriceps strength were assessed at four-time points: preoperative baseline, the morning (08:00–09:00) of POD1, the afternoon (16:00–17:00) of POD1, and the morning (08:00–09:00) of POD2. The quadriceps strength of both legs was tested using a hand-held dynamometer (Lafayette Manual Muscle Test System, Lafayette Instrument Company, Lafayette, Indiana, USA).13 Patients were instructed to extend their knees two times, with a 30 s pause between each attempt. The maximum force achieved was used in the analysis. The consumption of tramadol was converted to oral morphine equivalents.14 Preoperative data collection included the Hospital Anxiety and Depression Scale (HADS) ranging from 0 to 21. The probable presence of anxiety or depression was defined as a HADS anxiety or depression subscale score ≥11.15
Statistical analysis
We calculated the sample size required to detect a difference in NRS>3 among the three groups using pairwise comparison.5 Bonferroni correction was performed to adjust for the increase in type I error in multiple comparisons. Accordingly, 29 subjects were required in each group to achieve a statistical power of 90% at p value of <0.0167. Therefore, 32 patients were enrolled in each group to account for a 10% dropout rate. The Shapiro-Wilk and Kolmogorov-Smirnov tests were used to confirm normality of the data distribution. One-way analysis of variance and the Kruskal-Wallis test were used to analyze parametric and non-parametric continuous variables, respectively. Post-hoc analysis was performed using the t-test or Mann-Whitney test as appropriate and adjusted using the Bonferroni correction. Intergroup comparisons of categorical variables were conducted using Fisher’s exact test or the χ2 test as appropriate. Continuous variables are presented as mean±SD or median (IQR); categorical variables are presented as numbers (percentages). Statistical analyses were performed using R V.3.5.1 (R Foundation for Statistical Computing, Vienna, Austria), SPSS V.23.0, (IBM Corp., Armonk, New York, USA), or MedCalc Statistical Software V.18.11.3 (MedCalc Software Ltd., Ostend, Belgium). Statistical significance (p) was set at <0.05.
Results
Of the 102 patients screened for eligibility, 96 were enrolled in the study and randomly allocated to three groups: femoral triangle, proximal AC, or distal AC. One patient randomized to the proximal AC group experienced postoperative delirium (a disturbance in attention and awareness) and was withdrawn from the study during the postoperative follow-up period. Data from 95 patients were analyzed and patient flow through the study is shown in figure 2.
Consolidated Standards of Reporting Trials study flow diagram. AC, adductor canal.
Patient characteristics, operative data, and baseline assessments were similar among groups (table 1), as was the probable presence of depression or anxiety (p=0.157 and p=0.189, respectively). Leg length measurement based on radiograph showed no differences among groups. In the femoral triangle group, catheters were inserted 5.7±0.4 cm proximally from the proximal end of the AC. In the distal AC group, catheters were inserted 2.8±0.2 cm distally from the proximal end of the AC.
Demographic data and baseline assessments
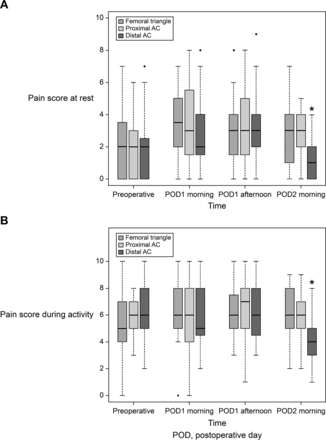
Postoperative pain scores are shown in figure 3. There was no significant difference in NRS pain scores at rest and during activity among the three groups in the morning and afternoon of POD1. NRS pain scores at rest (p=0.007) and during activity (p=0.005) were lower in the distal AC group in the morning of POD2 than those in the femoral triangle and proximal AC groups.
Pain scores. Boxplot represents the median with 25th/75th percentile. Whiskers reveal the minimum/maximum values, excluding outliers. Points represent the outliers. *P<0.05 between the distal AC and the other groups in the post-hoc analysis. AC, adductor canal.
Dynamometer readings and analgesia are shown in table 2. Quadriceps strength measurements in the operative and non-operative leg were not significantly different among groups in the morning and afternoon of POD1, and morning of POD2, respectively. Quadriceps strength measurements in the operated leg decreased from preoperative values in all groups at each time point. The cumulative amounts of 0.2% ropivacaine infused until 24, 36, and 48 hours postoperatively were comparable among groups. The number of patients who received rescue analgesics during the first 24 hours postoperatively was higher in the femoral triangle group than in the proximal and distal AC groups (p=0.027). However, cumulative opioid consumption was not significantly different among groups. There were no continuous block-related complications, such as falls, local anesthetic toxicity, or infection.
Quadriceps strength, perineural infusion pump data, and opioid consumption
Discussion
In this study, we found no difference in postoperative pain scores at rest and during activity in the morning and afternoon of POD1 after TKA among continuous femoral triangle, proximal AC, and distal AC blocks in the setting of periarticular local anesthetic infiltration. Among secondary outcomes, postoperative pain scores at rest and during activity in the morning of POD2 were significantly reduced in continuous distal ACB compared with continuous femoral triangle block and continuous proximal ACB.
The ideal location of continuous ACB after TKA may provide prolonged pain relief and improved functional recovery while preserving muscle strength. However, there are no clear recommendations for this approach due to inconsistent AC identification and different catheter insertion positions.4 5 9–11 Three studies showed no differences in analgesia between continuous femoral triangle block and ACB,4 9 10 whereas two others showed better analgesia or opioid-sparing effect in a proximal location than a distal location.5 11 In one study, pain score was lower on POD1 in the proximal ACB (midpoint of the thigh) than in the distal ACB (2–3 cm proximal to the adductor hiatus).5 In another, cumulative sufentanil consumption within 24 hours after TKA was lower in the proximal ACB (proximal end of AC) than in the middle ACB (3–5 cm distal to the proximal end of AC).11 Considering the most studied catheter positions,4 5 9–11 we selected three catheter insertion locations for continuous catheter analgesia: femoral triangle, proximal AC, and distal AC. In contrast to previous studies on ACBs,5 11 catheter insertion was performed postoperatively to minimize catheter tip displacement due to knee movement during surgery without tunneling technique. Our methodology is unique in the determination of each catheter insertion location in relation to individual femur length based on both ultrasound and radiographic images. This has the advantage of being an objective measurement compared with only ultrasound images or surface anatomy.
Our primary outcome showed comparable postoperative analgesia after TKA with continuous femoral triangle, proximal AC, and distal AC blocks. This finding is consistent with most studies on continuous catheter analgesia after TKA.4 9 10 Our results could be explained by the musculoaponeurotic tunnel feature of the AC and the target nerves of local anesthetic administration. The spread of local anesthetic is determined by the fascial limits and the muscles surrounding the space.16 17 The roof of the AC is a continuous fascia with thin proximal and thick distal portions.16 18 The thin superficial layer is the VAM, which connects the medial edge of the vastus medialis to the lateral edge of the adductor magnus, and the thick deep layer is the aponeurosis of the vastus medialis oblique.16 Because the femoral triangle is a space without VAM, longitudinal spread of local anesthetics may be less extensive than in AC.19 Moreover, the two thick layers of the roof in the narrow distal AC space may enhance local anesthetic spread as they are histologically aponeurotic and stiff.16 In a cadaveric study, 20 mL of dye injected into the distal femoral triangle stained the saphenous nerve and the nerve to vastus medialis in all specimens20; 10 mL of dye injected into the proximal end of the AC had spread to the saphenous nerve, posteromedial branch of the nerve to vastus medialis, superior medial genicular nerve, and genicular branch of the obturator nerve in all specimens.16 When 10 mL of dye was injected into an area 1–2 cm proximal to the adductor hiatus had spread into the popliteal fossa and stained the popliteal plexus, genicular branch of the posterior obturator nerve, as well as the saphenous nerve.21 However, 10–20 mL dye injected near the adductor hiatus led to varying staining results of the nerve to vastus medialis (0%–30%) because the nerve to vastus medialis is separated from the structures within the proximal AC by the fascia of the vastus medialis.16 20 21 However, the spread of dye injectate in cadavers may not be replicated in living subjects because of differences in tissue elasticity.16 17
Some secondary outcomes from our study differed from previous studies.4 5 9–11 Compared with a continuous femoral triangle block, continuous proximal and distal ACBs reduced rescue analgesic requirements during the first 24 hours. Analgesia on POD2 was improved with continuous distal ACB compared with continuous femoral triangle block and continuous proximal ACB. Theoretically, continuous administration of local anesthetics to the distal AC could spread into the popliteal fossa and contribute to analgesic effects on posterior knee pain. The addition of supplementary blocks targeting posterior knee pain to ACB has been shown to improve analgesic outcomes after TKA.22–24 Most previous ACB studies regarding have used periarticular local anesthetic infiltration.4 5 9 10 Among them, one study compared a distal ACB with a proximal location, but unlike ours, they investigated clinical outcomes up to POD1.5 At our institute, periarticular local anesthetic infiltration is a component of multimodal joint pathways and the posterior knee capsule was among the tissues routinely infiltrated. The analgesic benefit observed on POD2 may be attributed to the continuous distal ACB blocking posterior knee pain after the effect of periarticular local anesthetic infiltration disappeared.
There were no differences in quadriceps strength among groups, similar to previous studies of continuous femoral triangle blocks and ACBs.4 5 9–11 We found a significant decrease in quadriceps strength compared with preoperative baseline values in all groups. Postoperative quadriceps muscle strength can be affected by acute postoperative pain and surgical factors. This study was not powered to detect differences in this secondary outcome.
This study has several limitations. First, continuous catheter blocks were supplemented with periarticular local anesthetic infiltration, which might mask the analgesic benefit of distal ACB due to local anesthetic coverage for the posterior knee capsule on POD1. Second, the results apply to the specific local anesthetic type, basal rate, bolus volume, and lockout time used in this study. Third, more time was required to insert the catheter into the distal AC using a short-axis lateral to medial approach to avoid nerve damage by accurately tracing the saphenous nerve, which was not readily visible at this level. Finally, we determined each catheter insertion location in relation to individual femur length based on both ultrasound and radiographic images. However, whether the femur length ratio may be useful in determining the catheter insertion site in various races warrants further research.
In conclusion, continuous femoral triangle, proximal AC, and distal AC blocks provide comparable postoperative analgesia after TKA when periarticular local anesthetic infiltration is routinely administered. Quadriceps strength and opioid consumption did not differ among three continuous catheter blocks.
Supplemental material
Data availability statement
Data are available upon reasonable request.
Ethics statements
Patient consent for publication
Ethics approval
This study involves human participants and was approved by the Severance Hospital institutional review board on December 11, 2019 (protocol number: 4-2019-1038). This study was conducted in accordance with the principles of the Declaration of Helsinki. Participants gave informed consent to participate in the study before taking part.
Acknowledgments
The authors would like to thank Medical Illustration & Design, a part of the Medical Research Support Services of Yonsei University College of Medicine, for all the artistic support related to this work. The authors also would like to thank the biostatisticians employed for their statistical comments and analysis.
References
Supplementary materials
Supplementary Data
This web only file has been produced by the BMJ Publishing Group from an electronic file supplied by the author(s) and has not been edited for content.
Footnotes
Contributors BL and YSC contributed to study conception and design. BL, SJP, KKP, HJK, YSL, and YSC contributed to study conduct. BL and SJP contributed to data analysis. BL, SJP, KKP, and YSC contributed to manuscript preparation. YSC is a guarantor.
Funding This work was supported by a faculty research grant from the Yonsei University College of Medicine (grant number: 6-2020-0075).
Disclaimer The funder was not involved in the study.
Competing interests None declared.
Provenance and peer review Not commissioned; externally peer reviewed.
Supplemental material This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.